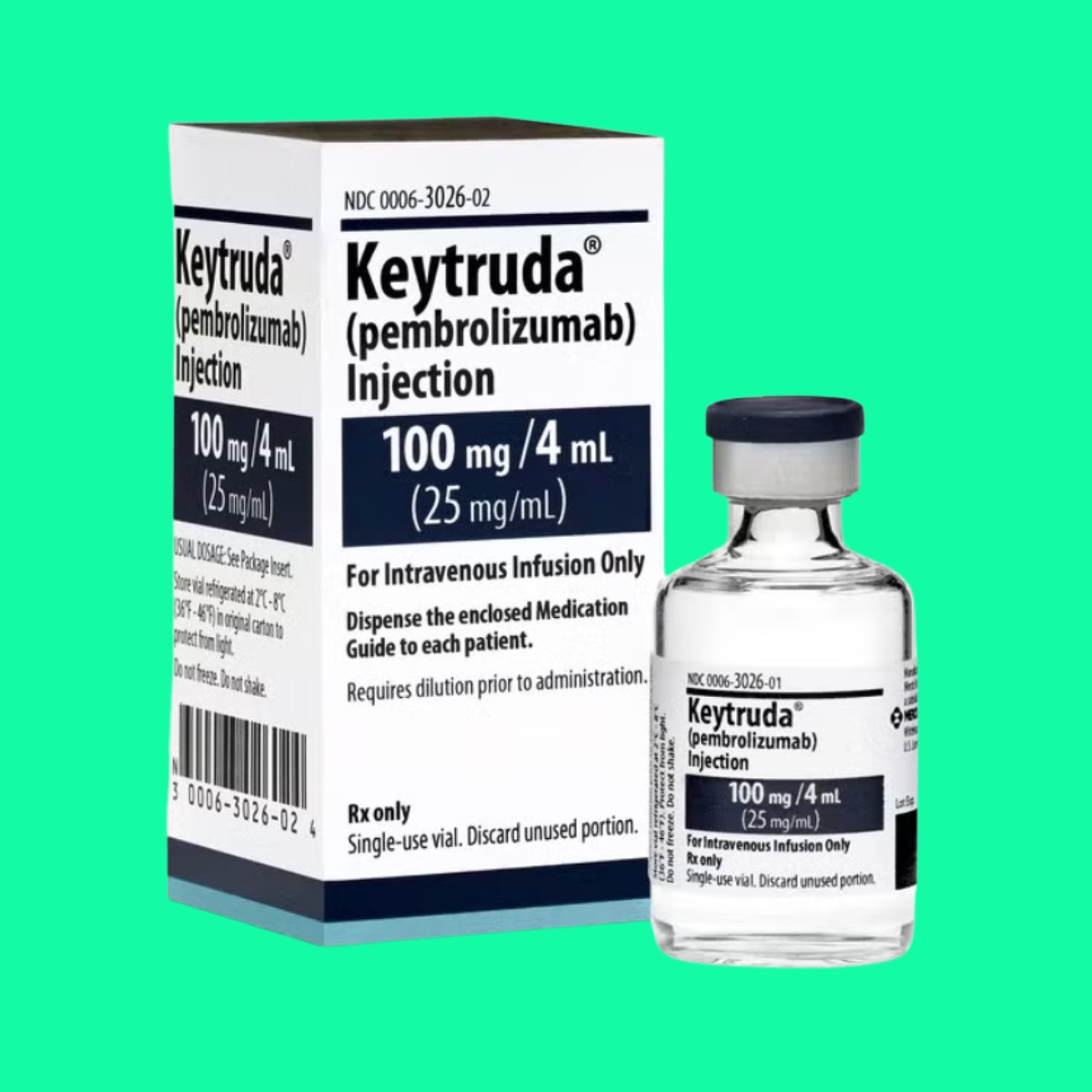
Description
What is Keytruda 100mg/4ml
Ingredient
Each 4ml of Keytruda 100mg/4ml contains the following ingredients:
- Pembrolizumab 100mg
- Excipients q.s.
Dosage form:
Present
Registration Number: QLSP-H02-1073-17
Packaging specifications: Box containing 1 bottle of 60 ml
Origin: Indonesia
Effects of Keytruda 100mg/4ml
Mechanism of action
Pembrolizumab is a monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with its ligands, PD-L1 and PD-L2. This binding leads to the activation of T-cell-mediated immune responses against tumor cells. Blocking PD-1 activity results in reduced tumor growth in mouse tumor models with similar genetic characteristics.
Pharmacokinetic characteristics
Absorption
Pembrolizumab is administered intravenously, has 100% bioavailability, and is not dependent on gastrointestinal absorption.
Distribution
The steady-state volume of distribution is approximately 7.7 L, primarily distributed within plasma and stromal spaces, and does not specifically bind to plasma proteins other than the target PD-1.
Metabolism
Pembrolizumab is broken down via proteolysis into amino acids and reused for the synthesis of other proteins.
Eliminate
The half-life is approximately 22–25 days.
What conditions is Keytruda 100mg/4ml indicated for?
Keytruda 100mg/4ml is indicated for:
- Treatment of inoperable or metastatic malignant melanoma, as well as in the context of adjuvant therapy after surgery for high-risk recurrence cases.
- Chemotherapy alone or in combination (with platinum or fluoropyrimidine as indicated) for patients with metastatic or inoperable non-small cell lung cancer.
- Monotherapy for patients with relapsed or refractory classic Hodgkin lymphoma who have failed autologous stem cell transplantation and brentuximab vedotin, or in cases ineligible for transplantation who have failed treatment with brentuximab vedotin.
- Monotherapy or combination therapy with enfortumab vedotin is indicated for patients with unresectable or metastatic ureteral/prostate carcinoma, as well as for urinary tract carcinoma when cystectomy is undesirable or not possible.
- Treatment of recurrent or metastatic squamous cell carcinoma of the head and neck with disease progression during or after platinum-based chemotherapy.
- Keytruda is used in combination with chemotherapy (with or without trastuzumab depending on HER2) for patients with progressive gastric or gastroesophageal shunt/metastatic PD-L1-positive disease.
- Monotherapy or combination chemotherapy (with/without bevacizumab) for recurrent/metastatic cervical cancer that is PD-L1 positive or has failed prior chemotherapy.
- Treatment of advanced hepatocellular carcinoma following prior systemic therapy (excluding PD-1/PD-L1 regimens).

Reviews
There are no reviews yet.